INTRODUCTION
Communications between the coronary arteries and the cardiac chambers (coronary-cameral fistulae) are rare forms of congenital heart disease. The sites of origin are the right coronary artery (55%), the left coronary artery (35%), or both coronary arteries (5%); the major termination sites are the right ventricle (40%), right atrium (RA) (26%), or pulmonary arteries (17%) [1]. Symptoms of angina or congestive heart failure may occur at the time of diagnosis but most present with subclinical or atypical symptoms. Therefore, the diagnosis is often delayed or incomplete unless multimodal imaging is used. We present an asymptomatic case of an unusual giant left circumflex coronary artery (LCx) aneurysm with fistulous communication towards the RA complicated with intraluminal thrombosis, all of which was fully delineated by multimodal cardiac imaging.
CASE
A 36-year-old man came to the family medicine clinic complaining of abdominal discomfort. After 3 days of gastrointestinal medications, the abdominal discomfort has gone, but there was a continuous murmur at the 3rd intercostal space left sternal border, which brought a suggestion of cardiac diseases such as patent ductus arteriosus, pulmonary valvular steno-regurgitation, or coronary arteriovenous fistulae. His blood pressure was 115/70 mmHg with a heart rate of 88 beats/min. His electrocardiogram showed a normal sinus rhythm with right-axis deviation without evidence of acute coronary syndrome. His simple chest X-ray showed a prominent pulmonary conus but was otherwise within normal limits. Laboratory tests were as follow: plasma brain natriuretic peptide, 16.4 pg/mL; total cholesterol, 273 mg/dL; low-density lipoprotein cholesterol, 208 mg/dL; high-density lipoprotein cholesterol, 43.0 mg/dL; and triglyceride, 203 mg/dL. He was an ex-smoker without any significant past medical history: no regular medications and no hypertension or diabetes mellitus.
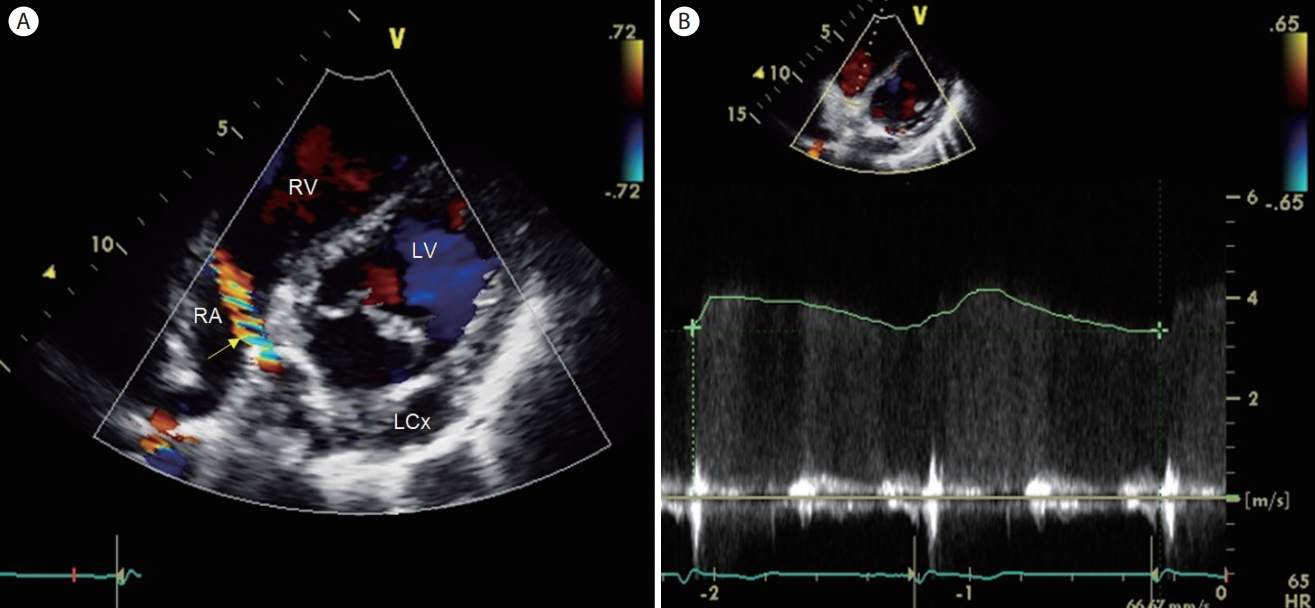
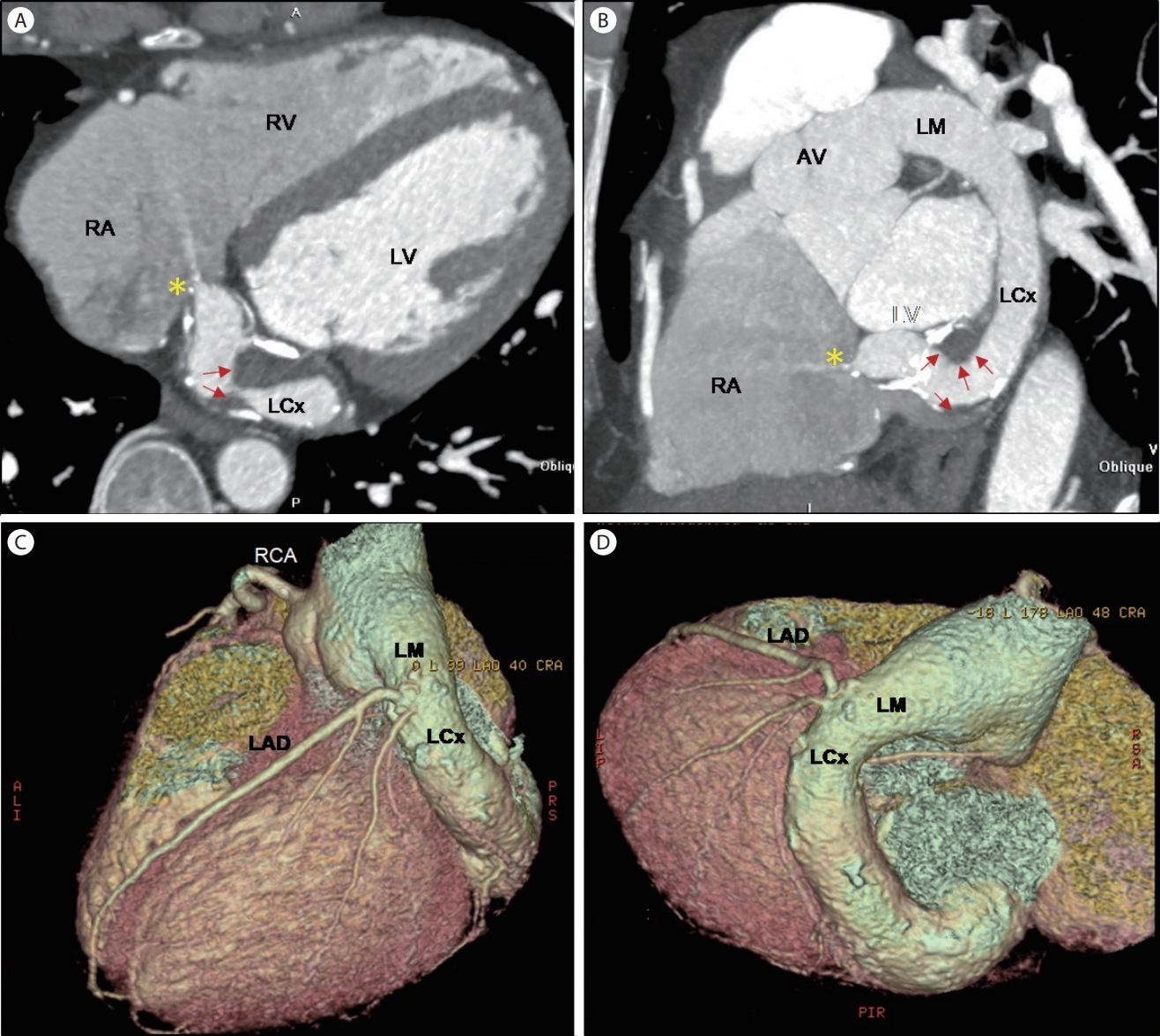
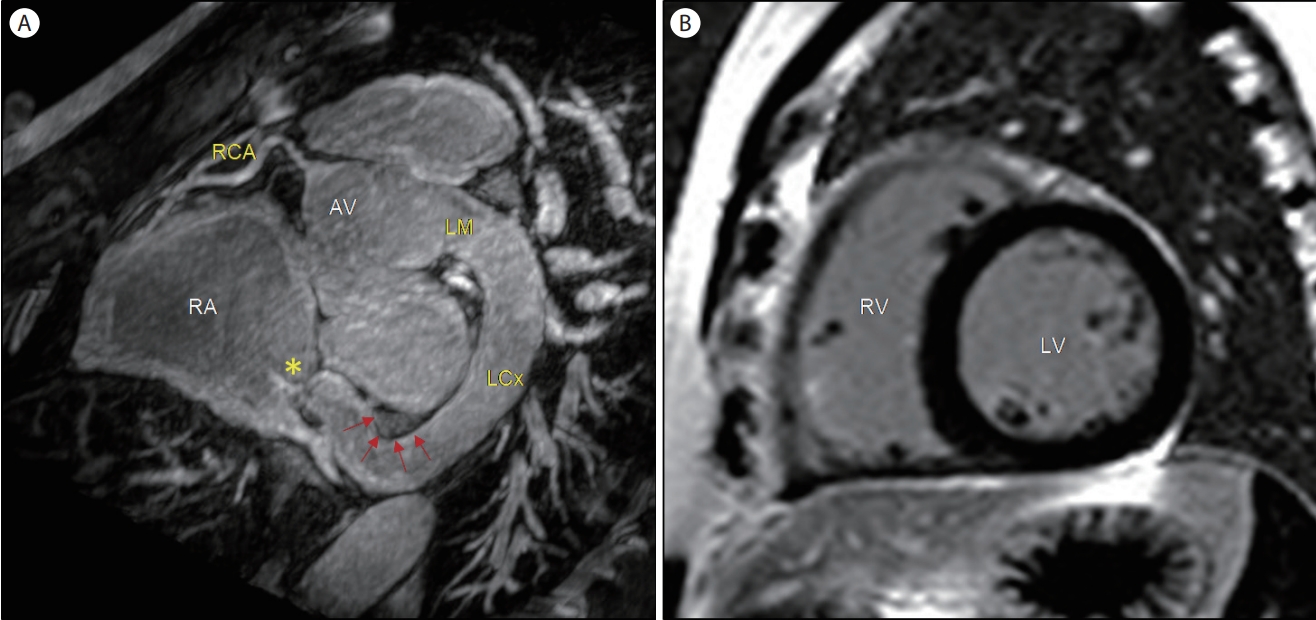
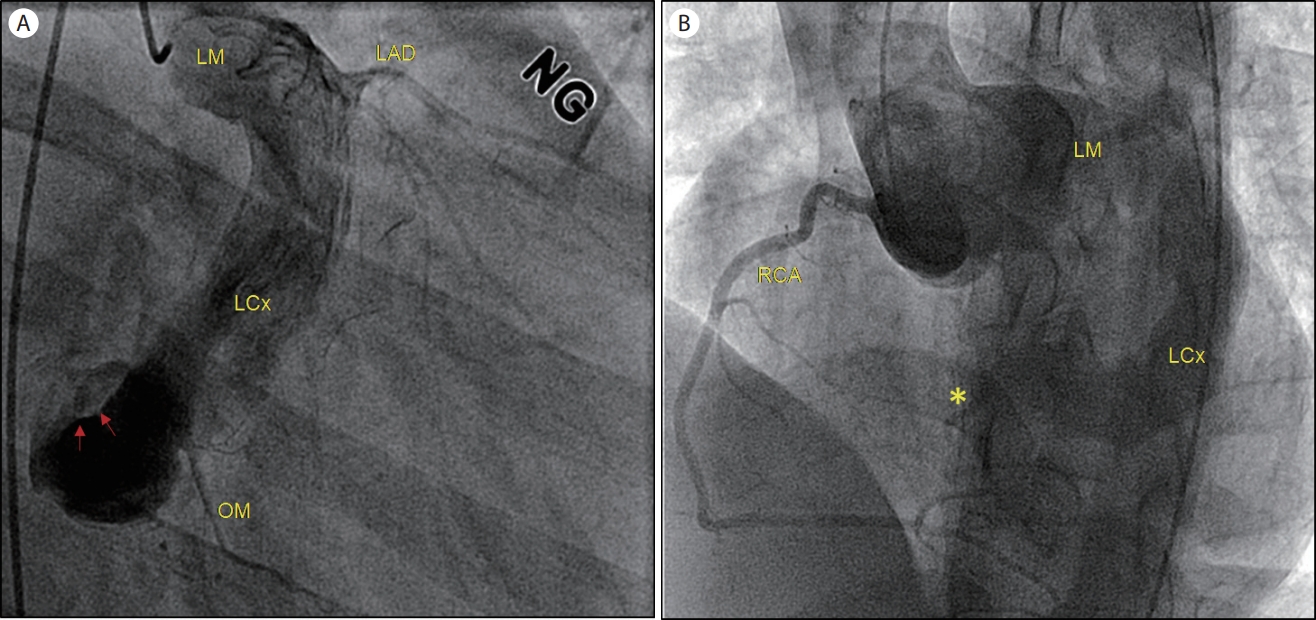
Referred to the cardiology clinic, non-invasive transthoracic echocardiography was performed to figure out the cause of the continuous murmur. There was no regional wall motion abnormality with normal left ventricular dimension and function (ejection fraction of 68%, normal diastology). The left main coronary artery ostium was markedly dilated, and the prominent LCx was noted in the routine 2D echo views (Fig. 1). In color-Doppler echo imaging, there was a continuous jet flow draining into the enlarged RA, presumptively from the dilated LCx, with a mean pressure gradient of 55.6 mmHg (Fig. 2). Subsequent coronary computed tomography angiography (CCTA) revealed a coronary artery anomaly: an LCx to RA fistula (diameter of 2.5 mm) and a coronary aneurysm (diameter of 20 mm) with calcified walls and intraluminal thrombi (Fig. 3). There was no stenosis in the right and left anterior descending coronary arteries. Cardiac magnetic resonance imaging (CMR) revealed no delayed hyper-enhancement of the left and right ventricular myocardium, and a Qp/Qs of 1.24 (Fig. 4). Finally, a coronary angiogram confirmed a giant coronary aneurysm from the left main trunk to the LCx with direct drainage to the RA through the distal small orifice (1.0-2.0 mm) (Fig. 5).
The 5-year follow-up was uneventful under aspirin and highdose statins, with a resolution of the intraluminal thrombi in the LCx but increased diameters of the coronary artery aneurysm (maximal diameter of 20 mm increasing to 24 mm) on the CCTA. He’s not complaining of any ischemic or heartrelated symptoms. Follow-up echocardiography showed no significant interval changes in the enlarged RA and estimated pulmonary arterial systolic pressure by trivial tricuspid regurgitation being 23 mmHg.
DISCUSSION
We described a case of a small coronary-cameral fistula (from the LCx to the RA) combined with a giant coronary aneurysm and intraluminal thrombi. Although most patients with coronary fistulae are asymptomatic, a hemodynamically significant left-to-right shunt may result in potential myocardial ischemia due to a coronary-steal phenomenon, possibly resulting in heart failure or pulmonary hypertension due to an excessive volume load on cardiac chambers [2]. Early anatomic correction may be considered when the shunt amount is significant (Qp/Qs > 1.5) to prevent late symptoms and complications [3]. In this case, an asymptomatic patient with a small-size cameral fistula having Qp/Qs of 1.24 by CMR, normal ventricular size and function without resting pulmonary hypertension warranted a medical follow-up.
Approximately 20-25% of coronary artery fistulae are associated with aneurysmal dilatation; the prevalence of a giant aneurysm (defined by a diameter of ≥ 2 cm) is only 0.02%, and spontaneous rupture is extremely rare [4,5]. This case showed an unusual giant aneurysm combined with intraluminal thrombi, resolved with antiplatelet medication. Because coronary artery aneurysms more than 3 cm in diameter are at risk of rupture, close CCTA monitoring of growth is essential [6]. Ultimately, elimination of a coronary artery fistula (either surgical or transcatheter) is recommended by the American Heart Association/American College of Cardiology when the fistula size is large or when small to moderate but with complications (myocardial ischemia, arrhythmias, ventricular dysfunction of unexplained etiology) [7].
For diagnosis, in this case, the auscultation of a loud continuous murmur played a key role in suspicion, but relevant multimodal cardiac images identified the coronary fistula and associated anomalies both anatomically and functionally. Transthoracic echocardiography is considered a proper initial imaging technique that can reveal enlarged coronary arteries or abnormal color-Doppler flow at the termination site of the shunt. Although it is difficult to delineate the entire course of the coronary anomaly via echo, echocardiography gave useful information regarding whether there were other associated cardiac lesions (for example, tetralogy of Fallot, patent ductus arteriosus, or an atrial septal defect) or corresponding structural or functional changes (chamber enlargement, myocardial dysfunction, or elevated intracardiac pressure) [2,8]. In this case, there was no other congenital anomaly and the terminal part of the shunt chamber (RA) was enlarged but there was no heart failure or significant resting pulmonary hypertension. The CCTA had the advantage of revealing the exact delineation of the origin and termination of the fistula through the entire course, including, in this case, an associated coronary aneurysm and intraluminal thrombi which were not defined by transthoracic echocardiography. CMR confirmed the presence of the fistula and added more information such as there are no areas of delayed hyper-enhancement suggestive of previous myocardial infarction and an estimate of the shunt amount (Qp/Qs). Ultimately the size and anatomical features of the fistula were reliably confirmed by invasive coronary angiography.
The present case highlights the importance of multimodal cardiac imaging in patients with a continuous murmur either to rule out a coronary fistula or to rule it in with exact delineation of the origin and termination as well as any complication [8].